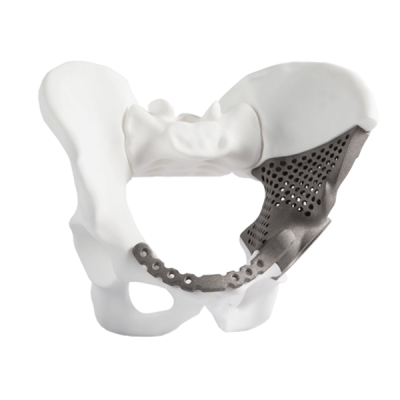
Instrumentaria offers over 3,500 registered products covering a wide range of orthopaedic and trauma implants and instruments for various surgical procedures. However, our extensive offer in some cases does not meet our customers’ needs due to a specific patient’s condition. For example, if due to a widespread tumour it is necessary to remove large portion of the pelvis, greater resection must be performed and the special design is often the only solution. Instrumentaria’s experts develop such products in collaboration with customers. The basis for design of each custom implant is surgeon’s request for such implant and CT scan to design precisely shaped bone that needs to be replaced. These implants are created in close collaboration with the operating surgeon with whom we analyse and fine-tune each and every feature of the design. After the doctor’s approval of the final design, the implant is produced using the additive manufacturing technology, popularly known as 3D print technology, which is a revolution in the production of medical implants because of its speed, accuracy and cost-effectiveness.
When working with Instrumentaria on designing custom implants, surgeons are in direct contact with the designers who are the main contact point for the clients through the entire process of development and production of the implant. This communication is an integral part of any successful design of a custom product that allows surgeons to review and discuss the CT data, the future design of the implant in real time.
The basis for each custom implant is surgeon’s requirement for the manufacture of product through Implant Prescription Form. This document can be downloaded and completed here.
The completed form is submitted to Instrumentaria, after which you will be contacted by the Research and Development Department to determine other parameters associated with the development itself, CT examination protocol and the method of further communication related to the design process.
-
Case studies
In the last two years, Instrumentaria has designed a number of personalized implants, and here you can see a brief overview of selected cases in order to realize the possibilities and advantages of this production methodology. Thanks to the precision and speed of additive technology, it is possible to reconstruct every segment of the human skeleton which is why personalized approach is used in orthopedic, trauma, cranial – maksiofacial and spinal surgery. By production of such implants, Instrumentaria is trying to offer its customers high-quality solutions for even the most complicated cases encountered, using the most advanced technological achievements.
Orthopedic Case Studies 2013/14
If you have any questions about custom implants, feel free to contact us via contact form.